PTC‑2105 for Sarcopenia & Sarcopenic Obesity
Lead MitoXcel™ geropeptide candidate designed to restore mitochondrial bioenergetics (ΔΨm) and reduce senescent‑cell inflammatory burden to improve physical function and body composition in older adults.

Program Overview
First‑in‑class ΔΨm‑targeted gerotherapeutic for age‑related loss of muscle mass and function.
- Indications: Sarcopenia and Sarcopenic Obesity
- Population: Older adults (≥ 60 years)
- Rationale: Aging and senescent cells exhibit reduced mitochondrial membrane potential (ΔΨm); restoring ΔΨm and removing senescent cells addresses upstream drivers of functional decline.
- Primary endpoints: Physical function improvement; favorable body composition (↓ fat, ↑ muscle)

Patient Need
Sarcopenia leads to loss of independence, increased falls, morbidity and mortality. In sarcopenic obesity, excess adiposity compounds disability risk. GLP‑1‑based pharmacotherapy can exacerbate lean mass loss, raising concerns for these patients.
- Loss of independence and increased healthcare utilization
- Higher risk of fractures, disability, and mortality
- Complex co‑morbid profile in sarcopenic obesity

Mechanism of Action
Two synergistic, mitochondrial‑mediated effects.
- ΔΨm Restoration in Aging Cells: Rapid improvement in mitochondrial efficiency, moving cells toward a youthful energetic phenotype.
- Selective Senescent Cell Apoptosis: Cells unable to restore ΔΨm—predominantly senescent cells—undergo apoptosis, reducing SASP cytokines (e.g., IL‑6, TNF‑α) and enabling muscle regeneration.
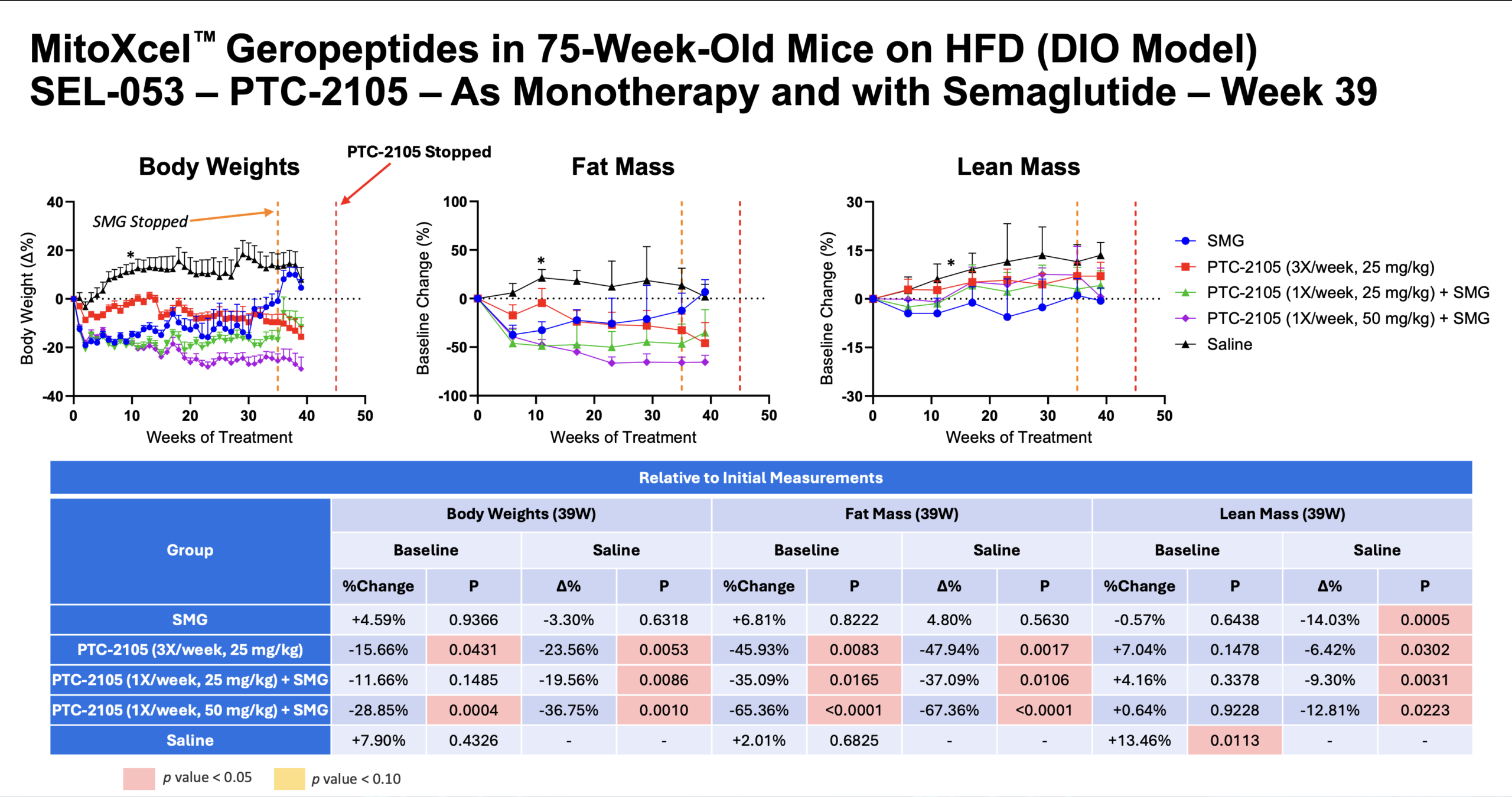
Preclinical studies have shown robust tissue‑wide effects without observed adverse events (illustrative summary).

Preclinical Evidence
Body Composition
- System‑wide senescent cell reduction via apoptosis
- Improved muscle regeneration and body composition
- No observed adverse effects in studies to date
Full datasets and protocols available under CDA.
Physical Function Benefit
- Rapid improvement in mitochondrial efficiency of aging cells
- Enhanced physical function metrics in aged animals
- No observed adverse effects in studies to date
Full datasets and protocols available under CDA.

Clinical Development Plan
Endpoints and eligibility aligned to functional benefit and body composition.
Primary Endpoints
- Physical function benefit (e.g., gait speed, 5xSTS, SPPB)
- Body composition improvement (loss of fat, gain of muscle)
Key Eligibility
- Age ≥ 60
- Sarcopenia or sarcopenic obesity diagnosis
- GLP‑1 agonist therapy not appropriate / contraindicated
To be finalized per protocol and regulatory guidance.


